pyani
Application and Python module for average nucleotide identity analyses of microbes.
pyani
We are deprecating pyani. Please use pyani-plus.
As part of the genomeRxiv project, we developed a new and improved implementation of average nucleotide identity methods that scales better on a wider range of computing environments. Please use pyani-plus in place of pyani.
You can download pyani-plus and find instructions for using it at the links below.
The pyani program and package has now reached the end of its useful life. It will no longer be maintained, and this repository will be deprecated.
I would like to take this opportunity to thank everyone who has used, worked on, or in any way improved pyani in its previous incarnations. It proved to be a popular and powerful software tool, used in over 1000 publications, and downloaded over 40,000 times. It’s been great, but it’s time to move on to better things (i.e. pyani-plus).
What happens next?
Hopefully, you become an active and enthusiastic user of pyani-plus.
We will be archiving this repository on 1st July 2025. At that point we will have closed all issues and pull requests, and the repository will become read-only to reflect its deprecated status.
What to do next
- I am happy using this version of
pyani- If you are happy using this version of
pyani, then by all means continue using it. But please be aware we will not be fixing bugs, or improving the codebase from this point on. The repository will be archived on 1st July 2025.
- If you are happy using this version of
pyaniis no longer working for me- There is a bug in
pyanithat I can’t live with - I would like to try the new improved
pyani-pluspyani-plusdoes the same job but is better in pretty much every relevant way, so please do visit the links below to download/install, and to read the documentation.
How issues will be affected
Any issues raised after 19th May 2025 will be cross-linked to the pyani-plus Issues page at https://github.com/pyani-plus/pyani-plus/issues and dealt with at that location
How pull requests will be affected
From 1st June 2025, no new pull requests will be merged other than in exceptional circumstances. Development has essentially ceased on this repository and we do not encourage late PRs.
But what if we don’t want pyani to die?
I’m sorry. pyani has had its time. If you would like to fork the project and maintain that fork, you are always free to do so. But I don’t think anyone wants pyani to live on any more than I do myself and, if I’m letting go, you may want to question whether using your finite time on this planet to keep this software alive is worth it.
Roll of Honour
We are grateful to all who have contributed to this software:
Contributors ✨ (emoji key):
(This project followed the all-contributors specification. Contributions of any kind welcome!)
Citing pyani
A complete guide to citing pyani is included in the file CITATIONS. Please cite the following manuscript in your work, if you have found pyani useful:
Pritchard et al. (2016) “Genomics and taxonomy in diagnostics for food security: soft-rotting enterobacterial plant pathogens” Anal. Methods 8, 12-24 DOI: 10.1039/C5AY02550H
Table of Contents
pyani
Overview
pyani is a software package and Python3 module that calculates average nucleotide identity (ANI) and related measures for whole genome comparisons, and renders relevant graphical summary output.
Where available, pyani can take advantage of multicore systems, and integrates with SGE/OGE-type job schedulers for the sequence comparisons.
pyani installs the program pyani, which enables command-line based analysis of genomes.
Installation
IMPORTANT NOTICE
THIS REPOSITORY DEFAULT BRANCH CONTAINS A DEVELOPMENT VERSION OF PYANI (v0.3+). IT HAS A DIFFERENT COMMAND-LINE INTERFACE THAN THE STABLE PYANI VERSION (v0.2.x).
Documentation for stable version pyani v0.2.x
IF YOU WISH TO INSTALL THE STABLE VERSION OF PYANI, PLEASE FOLLOW THESE INSTRUCTIONS FOR INSTALLING v0.2.x
The easiest way to install pyani v0.2 is to use conda or pip. conda is recommended for the simplest installation of third-party tool dependencies (mummer and BLAST/BLAST+).
conda
You will need to install the bioconda channel, following instructions at https://bioconda.github.io/user/install.html. Then, to create a new environment for pyani and install the program, issue the following command:
conda create --name pyani_env python=3.8 -y
conda activate pyani_env
conda install pyani
pip
pip will install pyani and its Python dependencies, but not the third-party tools.
pip3 install pyani
Third-party tools
Four alignment packages are required, to use all of pyani’s methods: mummer, BLAST+, legacy BLAST, and fastANI. (Note that it is not necessary to install all four tools to use pyani. You need only install the tools that are required for the method you wish to use.)
The simplest route to obtaining these tools is to use conda/bioconda:
conda install mummer blast legacy-blast fastani -y
But they can also be installed by following instructions from the tools’ own websites.
- BLAST+ (for
anib) ftp://ftp.ncbi.nlm.nih.gov/blast/executables/blast+/LATEST/ - legacy BLAST (for
aniblastall) ftp://ftp.ncbi.nlm.nih.gov/blast/executables/release/LATEST/ - MUMmer (for
anim) http://mummer.sourceforge.net/ - fastANI (for
fastani) https://github.com/ParBLiSS/FastANI
NOTE: Installing legacy BLAST
The legacy BLAST executable available from NCBI will not run on macOS Big Sur.
If you wish to use pyani blastall or the ANIblastall method with the legacy pyani interface, you will require a locally-installed copy of this package. This is one of the packages linked in the requirements-thirdparty.txt file.
Note: Installing fastANI
fastANI installation (via conda) will fail for Python 3.9 on macOS.
If you are using Python ≥3.9 to run pyani on macOS, fastANI will fail to install due to errors in the conda recipe (for build h0a26cfa_0). Using an earlier version of Python may be the simplest solution. However, you can also install fastANI yourself—via conda—using a slightly altered version of the command produced from the conda recipe, as shown below.
N.B. We have placed the fastANI requirement in its own requirements-fastani.txt file so that this will not also cause other installations to fail.
Bypassing conda
c++ -O3 -DNDEBUG -std=c++11 -Isrc \
-I ${ENV_DIR}/lib/include -mmacosx-version-min=10.7 \
-stdlib=libc++ -Xpreprocessor -fopenmp -lomp \
-DUSE_BOOST src/cgi/core_genome_identity.cpp -o fastANI \
${ENV_DIR}/lib/lib/libboost_math_c99.a \
-lstdc++ -lz -lm
Two alterations need to be made to the file name specified in the fifth line here:
- One of the
/libs should be removed. - The file extension should be changed from
.a, to.dylib.
The resultant command ought to look something like this (with ${ENV_DIR} being wherever you’re installing fastANI):
c++ -O3 -DNDEBUG -std=c++11 -Isrc \
-I ${ENV_DIR}/lib/include -mmacosx-version-min=10.7 \
-stdlib=libc++ -Xpreprocessor -fopenmp -lomp \
-DUSE_BOOST src/cgi/core_genome_identity.cpp -o fastANI \
${ENV_DIR}/lib/libboost_math_c99.dylib \
-lstdc++ -lz -lm
For a more technical overview of the issue, and other solutions that may be shared there, please see https://github.com/widdowquinn/pyani/issues/377.
Troubleshooting
This solution is how one of our developers managed to solve the problem. We hope it works for you, but can not guarantee this. Unfortunately, if this does not work for you, we will also not be able to troubleshoot the issue. In this instance, please file an issue at https://github.com/bioconda/bioconda-recipes.
Documentation (v0.3)
This README.md file provides a quick overview and walkthrough for THE DEVELOPMENT VERSION (v0.3+) OF pyani, and full documentation can be found at the link below:
THIS README AND THE DOCUMENTATION AT READTHEDOCS REFERS TO A DEVELOPMENT VERSION OF PYANI (v0.3+). IT HAS A DIFFERENT COMMAND-LINE INTERFACE THAN THE STABLE PYANI VERSION (v0.2.x).
THE STABLE VERSION OF PYANI (v0.2) DOES NOT HAVE THE pyani COMMAND
Older documentation (v0.2)
If you are using pyani v0.2.x, then please note that the command-line API has changed, and documentation for this version can be found at the following page:
Bugs, issues, problems and questions
If you would like to report a bug or problem with pyani, or ask a question of the developer(s), please raise an issue at the link below:
Walkthrough: A First Analysis
The command-line interface to pyani uses subcommands. These separate individual steps of an analysis into separate actions.
- Download genomes
- Create a database to hold genome data and analysis results
- Perform ANI analysis
- Report and visualise analysis results
- Generate species hypotheses (classify genomes) using the analysis results
The steps are described in detail with examples, below.
1. Download genome data
The first step is to obtain genome data for analysis. pyani expects to find each individual genome in its own FASTA file (that file can contain multiple sequences - chromosomes and plasmids; sequenced scaffolds, etc). All the FASTA files for an analysis are expected to be located in a single subdirectory (with optional labels and classes files). You can arrange your data manually, but pyani provides a subcommand that downloads all genomes in a taxon subtree from NCBI, and organises them ready for use with pyani.
We’ll use the pyani download subcommand to download all available genomes for Candidatus Blochmannia from NCBI. The taxon ID for this grouping is 203804.
pyani download -o C_blochmannia --email my.email@my.domain -t 203804 -v -l C_blochmannia_dl.log
The first argument is the output directory into which the downloaded genomes will be written (-o C_blochmannia). To download anything from NCBI we must provide an email address (--email my.email@my.domain), and to specify which taxon subtree we want to download we provide the taxon ID (-t 203804).
Here we also request verbose output (-v), and write a log file for reproducible research/diagnosing bugs and errors (-l C_blochmannia_dl.log).
This produces a new subdirectory (C_blochmannia) with the following contents:
$ tree C_blochmannia
C_blochmannia
├── GCF_000011745.1_ASM1174v1_genomic.fna
├── GCF_000011745.1_ASM1174v1_genomic.fna.gz
├── GCF_000011745.1_ASM1174v1_genomic.fna.md5
├── GCF_000011745.1_ASM1174v1_hashes.txt
├── GCF_000043285.1_ASM4328v1_genomic.fna
├── GCF_000043285.1_ASM4328v1_genomic.fna.gz
├── GCF_000043285.1_ASM4328v1_genomic.fna.md5
├── GCF_000043285.1_ASM4328v1_hashes.txt
├── GCF_000185985.2_ASM18598v2_genomic.fna
├── GCF_000185985.2_ASM18598v2_genomic.fna.gz
├── GCF_000185985.2_ASM18598v2_genomic.fna.md5
├── GCF_000185985.2_ASM18598v2_hashes.txt
├── GCF_000331065.1_ASM33106v1_genomic.fna
├── GCF_000331065.1_ASM33106v1_genomic.fna.gz
├── GCF_000331065.1_ASM33106v1_genomic.fna.md5
├── GCF_000331065.1_ASM33106v1_hashes.txt
├── GCF_000973505.1_ASM97350v1_genomic.fna
├── GCF_000973505.1_ASM97350v1_genomic.fna.gz
├── GCF_000973505.1_ASM97350v1_genomic.fna.md5
├── GCF_000973505.1_ASM97350v1_hashes.txt
├── GCF_000973545.1_ASM97354v1_genomic.fna
├── GCF_000973545.1_ASM97354v1_genomic.fna.gz
├── GCF_000973545.1_ASM97354v1_genomic.fna.md5
├── GCF_000973545.1_ASM97354v1_hashes.txt
├── classes.txt
└── labels.txt
Seven genomes have been downloaded, and each is represented by four files:
_genomic.fna.gz: the compressed genome sequence_genomic.fna: the uncompressed genome sequence_genomic.fna.md5: an MD5 hash/checksum of the (uncompressed) genome sequence; this was generated during the download_hashes.txt: a list of MD5 hashes; this is provided by NCBI and is a reference to be sure that the download did not corrupt the genome sequence
There are two additional plain text files: classes.txt and labels.txt, which provide alternative labels for use in the analysis. These files are generated during the download.
2. Create an analysis database
pyani uses a database to store genome data and analysis results. This is convenient for data sharing and developing custom analyses, but also makes it easier to extend an existing ANI analysis with new genomes, without having to repeat calculations that were already performed.
To create a new, clean, database in the default location (.pyani/pyanidb) issue the command:
pyani createdb -v -l C_blochmannia_createdb.log
As above, the verbose (-v) and log file (-l C_blochmannia_createdb.log) options allow for reproducible work. The default database location is in the hidden directory (.pyani):
$ tree .pyani
.pyani
└── pyanidb
Subsequent pyani commands will assume this location for the database, but you can specify the location when creating a database, or using an existing database.
3. Conduct ANI analysis
pyani provides four subcommands to run ANI analyses:
anim: ANImanib: ANIb, using BLAST+aniblastall: ANIb, using legacy BLASTtetra: TETRA
In this walkthrough, we’ll run ANIm on the downloaded genomes, using the command:
pyani anim -i C_blochmannia -o C_blochmannia_ANIm -v -l C_blochmannia_ANIm.log \
--name "C. blochmannia run 1" \
--labels C_blochmannia/labels.txt --classes C_blochmannia/classes.txt
All four analysis commands operate in a similar way. The first two arguments are paths to directories: the first path is to a directory containing input genomes, and the second is the path to an output directory for storing intermediate results. The -v and -l arguments work as above, specifying verbose output and logging output to a file.
You will probably notice that the verbose output is very verbose, to enable informative identification of any problems. In particular, the verbose output (which is also written to the log file) writes out the command-lines used for the pairwise comparisons so, if something goes wrong, you can test whether a specific comparison can be run at the command-line at all, to aid diagnosis of any problems.
Rerunning the same analysis
One reason for using a database backend for analysis results is so that, for very large analyses, we do not ever need to recalculate a pairwise genome comparison. All the analysis subcommands check whether input genomes have been used before (using the unique MD5 hash for each genome to identify whether it’s been used previously), and whether the comparison of two genomes has been run, with the particular analysis settings that were used. If either genome was not seen before, or if the analysis settings are different, the comparison is performed.
You can test this for yourself by running the analysis command again, as below. You will see a number of messages indicating that genomes have been seen before, and that analyses performed before were skipped:
$ pyani anim -i C_blochmannia -o C_blochmannia_ANIm -v -l C_blochmannia_ANIm.log \
--name "C. blochmannia run 2" \
--labels C_blochmannia/labels.txt --classes C_blochmannia/classes.txt
INFO: command-line: pyani anim -i C_blochmannia -o C_blochmannia_ANIm -v -l C_blochmannia_ANIm.log
INFO: Running ANIm analysis
INFO: Adding analysis information to database .pyani/pyanidb
INFO: Current analysis has ID 2 in this database
INFO: Identifying input genome/hash files:
[…]
INFO: Adding genome data to database...
WARNING: Genome already in database with this hash and path!
WARNING: Using existing genome from database, row 1
[…]
INFO: Complete pairwise comparison list:
[(1, 2), (1, 3), (1, 4), (1, 5), (1, 6), (2, 3), (2, 4), (2, 5), (2, 6), (3, 4), (3, 5), (3, 6), (4, 5), (4, 6), (5, 6)]
INFO: Excluding pre-calculated comparisons
INFO: Comparisons still to be performed:
[]
INFO: All comparison results already present in database (skipping comparisons)
INFO: Completed. Time taken: 0.211
4. Reporting Analyses and Analysis Results
Once an analysis is run, the results are placed in a local SQLite database, which can be queried for information about the analyses that have been run. You can request information about:
--runs: show all analysis runs with results stored in the database--runs_genomes: show all the analysis runs with results in the database, and all the genomes analysed in each run--genomes: show all the genomes used for any analysis in the database--genomes_runs: for each genome in the database, also list the analysis results it participates in--run_results: show all the pairwise comparison results for a named run (run IDs can be obtained with the--runsargument
The report tables are written to a named directory (compulsory argument), and are written by default to a .tab plain-text format, but HTML and Excel format can also be requested with the --formats argument:
$ pyani report -v --runs -o C_blochmannia_ANIm/ --formats html,excel,stdout
INFO: Processed arguments: Namespace(cmdline='./pyani report -v --runs -o C_blochmannia_ANIm/ --formats html,excel', dbpath='.pyani/pyanidb', formats='html,excel', func=<function subcmd_report at 0x10c674a60>, logfile=None, outdir='C_blochmannia_ANIm/', run_results=False, show_genomes=False, show_genomes_runs=False, show_runs=True, show_runs_genomes=False, verbose=True)
INFO: command-line: ./pyani report -v --runs -o C_blochmannia_ANIm/ --formats html,excel
INFO: Creating output in formats: ['excel', 'tab', 'html']
INFO: Using database: .pyani/pyanidb
INFO: Writing table of pyani runs from the database to C_blochmannia_ANIm/runs.*
INFO: Completed. Time taken: 0.937
$ tree -L 1 C_blochmannia_ANIm/
C_blochmannia_ANIm/
├── nucmer_output
├── runs.html
├── runs.tab
└── runs.xlsx
To see all of the pairwise results for an individual run, the run ID must be provided. It is possible to get results for more than one run ID by providing a comma-separated list of run IDs (though each run’s results will be provided in a separate file):
$ pyani report -v --runs -o C_blochmannia_ANIm/ --formats html,excel --run_results 1,2,3,4
INFO: Processed arguments: Namespace(cmdline='./pyani report -v --runs -o C_blochmannia_ANIm/ --formats html,excel --run_results 1,2,3,4', dbpath='.pyani/pyanidb', formats='html,excel', func=<function subcmd_report at 0x108616a60>, logfile=None, outdir='C_blochmannia_ANIm/', run_results='1,2,3,4', show_genomes=False, show_genomes_runs=False, show_runs=True, show_runs_genomes=False, verbose=True)
INFO: command-line: ./pyani report -v --runs -o C_blochmannia_ANIm/ --formats html,excel --run_results 1,2,3,4
INFO: Creating output in formats: ['tab', 'excel', 'html']
INFO: Using database: .pyani/pyanidb
INFO: Writing table of pyani runs from the database to C_blochmannia_ANIm/runs.*
INFO: Attempting to write results tables for runs: ['1', '2', '3', '4']
INFO: Collecting data for run with ID: 1
INFO: Collecting data for run with ID: 2
INFO: Collecting data for run with ID: 3
INFO: Collecting data for run with ID: 4
INFO: Completed. Time taken: 1.285
You can see a run’s results in the terminal by specifying the stdout format. For example, to see the identity, coverage, and other output matrices, you would specify --run_matrices <RUN> and --formats=stdout as below:
$ pyani report -o C_blochmannia_ANIm --formats=stdout --run_matrices 1
TABLE: C_blochmannia_ANIm/matrix_identity_1
C. Blochmannia pennsylvanicus BPEN C. Blochmannia floridanus C. Blochmannia vafer BVAF C. Blochmannia chromaiodes 640 B. endosymbiont of Polyrhachis (Hedomyrma) turneri 675 B. endosymbiont of Camponotus (Colobopsis) obliquus 757
C. Blochmannia pennsylvanicus BPEN 1.000000 0.834866 0.836903 0.980244 0.843700 0.829509
C. Blochmannia floridanus 0.834866 1.000000 0.828733 0.834916 0.847060 0.857859
C. Blochmannia vafer BVAF 0.836903 0.828733 1.000000 0.837811 0.866015 0.844438
C. Blochmannia chromaiodes 640 0.980244 0.834916 0.837811 1.000000 0.849834 0.834769
B. endosymbiont of Polyrhachis (Hedomyrma) turn... 0.843700 0.847060 0.866015 0.849834 1.000000 0.844228
B. endosymbiont of Camponotus (Colobopsis) obli... 0.829509 0.857859 0.844438 0.834769 0.844228 1.000000
TABLE: C_blochmannia_ANIm/matrix_coverage_1
C. Blochmannia pennsylvanicus BPEN C. Blochmannia floridanus C. Blochmannia vafer BVAF C. Blochmannia chromaiodes 640 B. endosymbiont of Polyrhachis (Hedomyrma) turneri 675 B. endosymbiont of Camponotus (Colobopsis) obliquus 757
C. Blochmannia pennsylvanicus BPEN 1.000000 0.045736 0.041404 1.000306 0.017263 0.021027
C. Blochmannia floridanus 0.051317 1.000000 0.152609 0.054930 0.016366 0.010749
C. Blochmannia vafer BVAF 0.045362 0.149012 1.000000 0.046520 0.008356 0.014706
C. Blochmannia chromaiodes 640 1.000856 0.048983 0.042485 1.000000 0.014056 0.016140
B. endosymbiont of Polyrhachis (Hedomyrma) turn... 0.018238 0.015410 0.008058 0.014841 1.000000 0.020416
B. endosymbiont of Camponotus (Colobopsis) obli... 0.021508 0.009799 0.013730 0.016500 0.019766 1.000000
TABLE: C_blochmannia_ANIm/matrix_aln_lengths_1
C. Blochmannia pennsylvanicus BPEN C. Blochmannia floridanus C. Blochmannia vafer BVAF C. Blochmannia chromaiodes 640 B. endosymbiont of Polyrhachis (Hedomyrma) turneri 675 B. endosymbiont of Camponotus (Colobopsis) obliquus 757
C. Blochmannia pennsylvanicus BPEN 791654.0 36207.0 32778.0 791896.0 13666.0 16646.0
C. Blochmannia floridanus 36207.0 705557.0 107674.0 38756.0 11547.0 7584.0
C. Blochmannia vafer BVAF 32778.0 107674.0 722585.0 33615.0 6038.0 10626.0
C. Blochmannia chromaiodes 640 791896.0 38756.0 33615.0 791219.0 11121.0 12770.0
B. endosymbiont of Polyrhachis (Hedomyrma) turn... 13666.0 11547.0 6038.0 11121.0 749321.0 15298.0
B. endosymbiont of Camponotus (Colobopsis) obli... 16646.0 7584.0 10626.0 12770.0 15298.0 NaN
TABLE: C_blochmannia_ANIm/matrix_sim_errors_1
C. Blochmannia pennsylvanicus BPEN C. Blochmannia floridanus C. Blochmannia vafer BVAF C. Blochmannia chromaiodes 640 B. endosymbiont of Polyrhachis (Hedomyrma) turneri 675 B. endosymbiont of Camponotus (Colobopsis) obliquus 757
C. Blochmannia pennsylvanicus BPEN 0.0 5979.0 5346.0 15645.0 2136.0 2838.0
C. Blochmannia floridanus 5979.0 0.0 18441.0 6398.0 1766.0 1078.0
C. Blochmannia vafer BVAF 5346.0 18441.0 0.0 5452.0 809.0 1653.0
C. Blochmannia chromaiodes 640 15645.0 6398.0 5452.0 0.0 1670.0 2110.0
B. endosymbiont of Polyrhachis (Hedomyrma) turn... 2136.0 1766.0 809.0 1670.0 0.0 2383.0
B. endosymbiont of Camponotus (Colobopsis) obli... 2838.0 1078.0 1653.0 2110.0 2383.0 0.0
TABLE: C_blochmannia_ANIm/matrix_hadamard_1
C. Blochmannia pennsylvanicus BPEN C. Blochmannia floridanus C. Blochmannia vafer BVAF C. Blochmannia chromaiodes 640 B. endosymbiont of Polyrhachis (Hedomyrma) turneri 675 B. endosymbiont of Camponotus (Colobopsis) obliquus 757
C. Blochmannia pennsylvanicus BPEN 1.000000 0.038183 0.034652 0.980543 0.014564 0.017442
C. Blochmannia floridanus 0.042843 1.000000 0.126472 0.045862 0.013863 0.009221
C. Blochmannia vafer BVAF 0.037964 0.123491 1.000000 0.038975 0.007237 0.012418
C. Blochmannia chromaiodes 640 0.981082 0.040896 0.035594 1.000000 0.011945 0.013473
B. endosymbiont of Polyrhachis (Hedomyrma) turn... 0.015387 0.013053 0.006978 0.012613 1.000000 0.017236
B. endosymbiont of Camponotus (Colobopsis) obli... 0.017841 0.008406 0.011594 0.013774 0.016687 1.000000
5. Generating graphical output for ANI
The output of a pyani run can also be represented graphically, using the plot subcommand. For example, the command:
pyani plot -o C_blochmannia_ANIm --run_id 1 -v --formats png,pdf
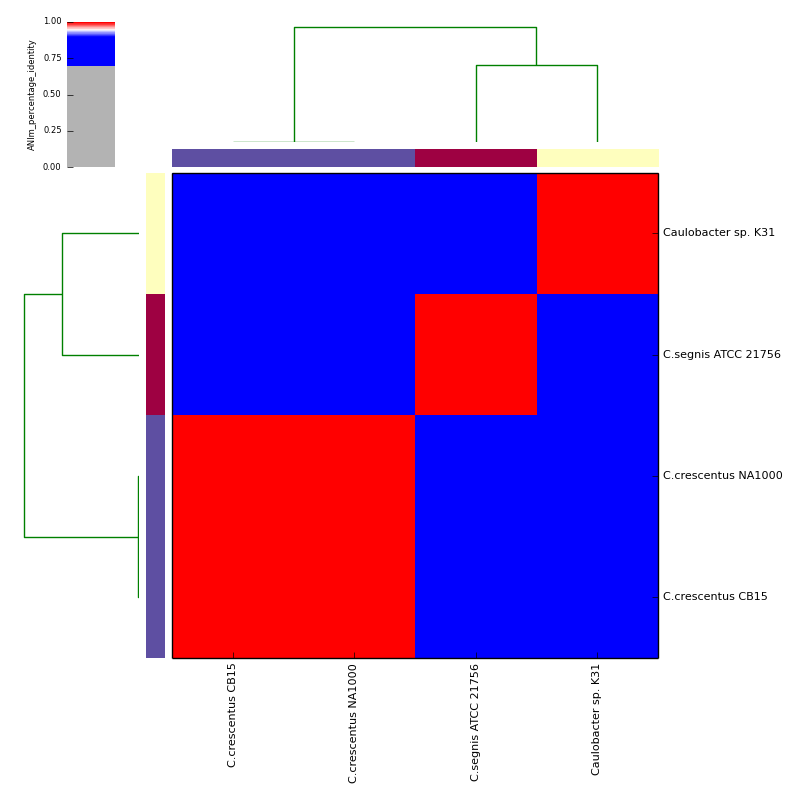
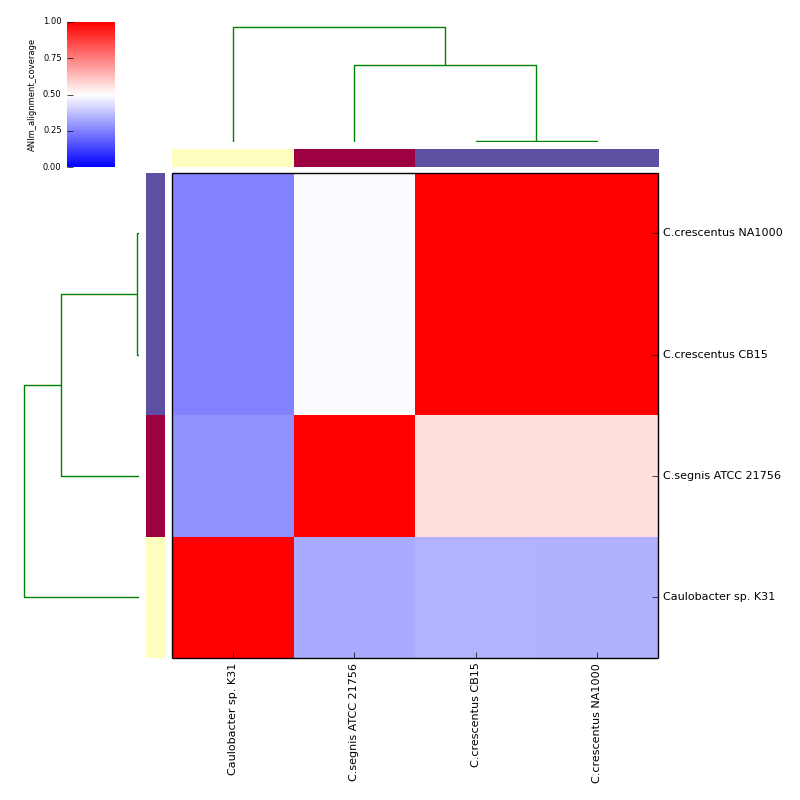
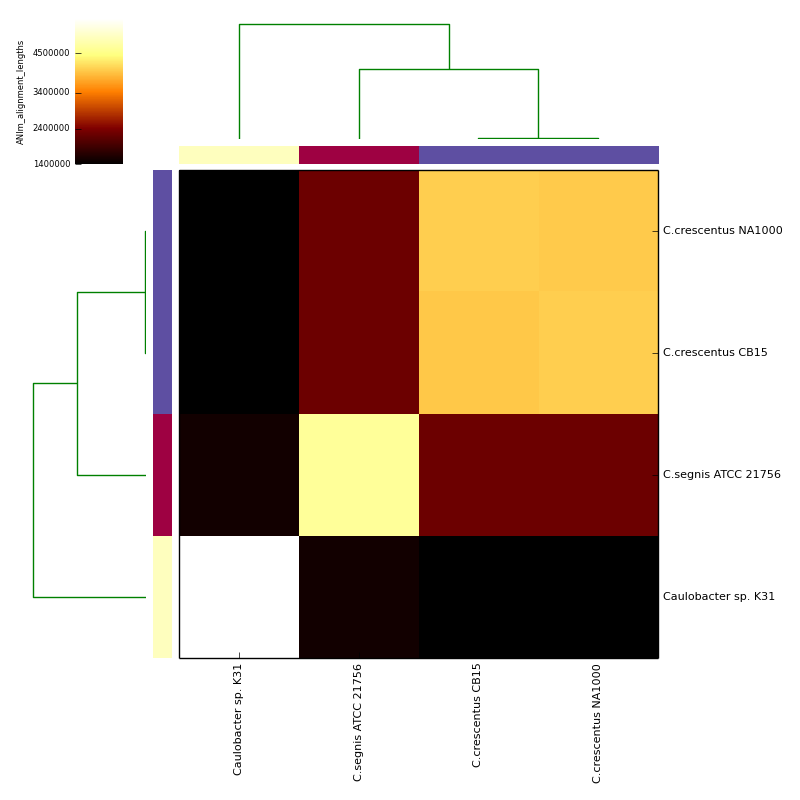
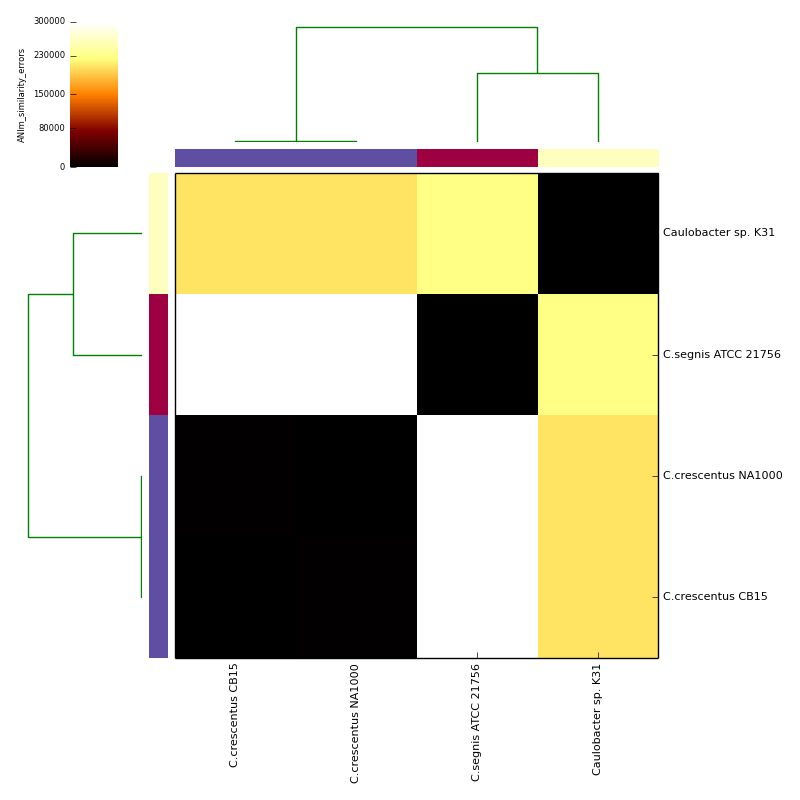
will place .pdf and .png format output in the C_blochmannia_ANIm output directory for the run with ID 1, generated above. Five heatmaps are generated:
- percentage identity
- percentage coverage (for both query and subject)
- alignment length (total aligned length)
- similarity errors (total number of mismatches, not including indels)
- hadamard matrix (dot product of identity and coverage matrices)
The heatmaps also include dendrograms, clustering the rows and columns by overall similarity.
NOTE: Running pyani plot with a large number of genomes (~500) and the default figure output (--method seaborn) may reduce output figure quality:
- labels in
.pngfiles may be difficult to read .pdffiles may render very slowly due to the large number of numerical values in heatmap cells.
With large datasets, --method mpl (matplotlib) is recommended.
Please be aware that the matrix orientation differs for these two options; so, with seaborn (the default, --method seaborn), the orientation of self-comparisons is top left to bottom right (\), while with matplotlib (--method mpl) the orientation is bottom left to top right (/).
6. Classifying Genomes from Analysis Results
Using a scheduler
SGE/OGE
The --scheduler SGE argument allows one to use pyani with an an SGE-type scheduler.
In order for this work, one must be able to submit jobs using the qsub command. By default, this will batch the pairwise comparisons in array jobs of 10,000, in order to avoid clogging the scheduler queue. Each comparison will be run as a single-core task in an array job.
Arguments assigned by Pyani
The following arguments will be automatically set:
-N job_name # this is the value passed to `--name`
-cwd
-o ./stdout # cwd/ + "stdout"
-e ./stderr # cwd/ + "stderr"
Modifiable arguments
The number of pairwise comparisons submitted per chunk can be modified using:
--SGEgroupsize *number*
The job prefix to use can be modified using:
--jobprefix *prefix*
Specifying additional arguments
Additional SGE arguments may be specified with:
--SGEargs "<your arguments here>"
Running pyani version 0.2.x
Script: average_nucleotide_identity.py
The average_nucleotide_identity.py script - installed as part of this package - enables straightforward ANI analysis at the command-line, and uses the pyani module behind the scenes.
You can get a summary of available command-line options with average_nucleotide_identity.py -h
$ ./average_nucleotide_identity.py -h
usage: average_nucleotide_identity.py [-h] [-o OUTDIRNAME] [-i INDIRNAME] [-v]
[-f] [-s FRAGSIZE] [-l LOGFILE]
[--skip_nucmer] [--skip_blastn]
[--noclobber] [--nocompress] [-g]
[--gformat GFORMAT] [--gmethod GMETHOD]
[--labels LABELS] [--classes CLASSES]
[-m METHOD] [--scheduler SCHEDULER]
[--workers WORKERS]
[--SGEgroupsize SGEGROUPSIZE]
[--maxmatch] [--nucmer_exe NUCMER_EXE]
[--blastn_exe BLASTN_EXE]
[--makeblastdb_exe MAKEBLASTDB_EXE]
[--blastall_exe BLASTALL_EXE]
[--formatdb_exe FORMATDB_EXE]
[--write_excel] [--subsample SUBSAMPLE]
[--seed SEED] [--jobprefix JOBPREFIX]
[…]
Example data and output can be found in the directory test_ani_data. The data are chromosomes of four isolates of Caulobacter. Basic analyses can be performed with the command lines:
./average_nucleotide_identity.py -i tests/test_ani_data/ -o tests/test_ANIm_output -m ANIm -g
./average_nucleotide_identity.py -i tests/test_ani_data/ -o tests/test_ANIb_output -m ANIb -g
./average_nucleotide_identity.py -i tests/test_ani_data/ -o tests/test_ANIblastall_output -m ANIblastall -g
./average_nucleotide_identity.py -i tests/test_ani_data/ -o tests/test_TETRA_output -m TETRA -g
The graphical output below, supporting assignment of NC_002696 and NC_011916 to the same species (C.crescentus), and the other two isolates to distinct species (NC_014100:C.segnis; NC_010338:C. sp K31), was generated with the command-line:
./average_nucleotide_identity.py -v -i tests/test_ani_data/ \
-o tests/test_ANIm_output/ -g --gformat png,pdf,eps \
--classes tests/test_ani_data/classes.tab \
--labels tests/test_ani_data/labels.tab
Script: genbank_get_genomes_by_taxon.py
The script genbank_get_genomes_by_taxon.py, installed by this package, enables download of genomes from NCBI, specified by taxon ID. The script will download all available assemblies for taxa at or below the specified node in the NCBI taxonomy tree.
Command-line options can be viewed using:
$ genbank_get_genomes_by_taxon.py -h
usage: genbacnk_get_genomes_by_taxon.py [-h] [-o OUTDIRNAME] [-t TAXON] [-v]
[-f] [--noclobber] [-l LOGFILE]
[--format FORMAT] [--email EMAIL]
[--retries RETRIES]
[--batchsize BATCHSIZE]
[…]
For example, the NCBI taxonomy ID for Caulobacter is 75, so all publicly-available Caulobacter sequences can be obtained using the command-line:
$ genbank_get_genomes_by_taxon.py -o Caulobacter_downloads -v -t 75 -l Caulobacter_downloads.log --email me@my.email.domain
INFO: genbank_get_genomes_by_taxon.py: Mon Apr 18 17:22:54 2016
INFO: command-line: /Users/lpritc/Virtualenvs/pyani3/bin/genbank_get_genomes_by_taxon.py -o Caulobacter_downloads -v -t 75 -l Caulobacter_downloads.log --email me@my.email.domain
INFO: Namespace(batchsize=10000, email='me@my.email.domain', force=False, format='gbk,fasta', logfile='Caulobacter_downloads.log', noclobber=False, outdirname='Caulobacter_downloads', retries=20, taxon='75', verbose=True)
INFO: Set NCBI contact email to me@my.email.domain
INFO: Creating directory Caulobacter_downloads
INFO: Output directory: Caulobacter_downloads
INFO: Passed taxon IDs: 75
INFO: Entrez ESearch with query: txid75[Organism:exp]
INFO: Entrez ESearch returns 29 assembly IDs
INFO: Identified 29 unique assemblies
INFO: Taxon 75: 29 assemblies
[…]
INFO: Assembly 639581: 271 contigs
INFO: Assembly 233261: 17 contigs
INFO: Assembly 575291: 48 contigs
INFO: Mon Apr 18 17:25:46 2016
INFO: Done.
NOTE: You must provide a valid email to identify yourself to NCBI for troubleshooting.
The number of attempted retries for each download, and the size of a batch download can be modified. By default, the script will attempt 20 download retries, and obtain sequences in batches of 10,000.
Method and Output Description
Average Nucleotide Identity (ANI)
This module calculates Average Nucleotide Identity (ANI) according to one of a number of alternative methods described in, e.g.
- Richter M, Rossello-Mora R (2009) Shifting the genomic gold standard for the prokaryotic species definition. Proc Natl Acad Sci USA 106: 19126-19131. doi:10.1073/pnas.0906412106. (ANI1020, ANIm, ANIb)
- Goris J, Konstantinidis KT, Klappenbach JA, Coenye T, Vandamme P, et al. (2007) DNA-DNA hybridization values and their relationship to whole-genome sequence similarities. Int J Syst Evol Micr 57: 81-91. doi:10.1099/ijs.0.64483-0.
ANI is proposed to be the appropriate in silico substitute for DNA-DNA hybridisation (DDH), and so useful for delineating species boundaries. A typical percentage threshold for species boundary in the literature is 95% ANI (e.g. Richter et al. 2009).
All ANI methods follow the basic algorithm:
- Align the genome of organism 1 against that of organism 2, and identify the matching regions.
- Calculate the percentage nucleotide identity of the matching regions, as an average for all matching regions.
Methods differ on: (1) what alignment algorithm is used, and the choice of parameters (this affects the aligned region boundaries); (2) what the input is for alignment (typically either fragments of fixed size, or the most complete assembly available).
- ANIm: uses MUMmer (NUCmer) to align the input sequences
- ANIb: uses BLASTN+ to align 1020nt fragments of the input sequences
- ANIblastall: uses legacy BLASTN to align 10al20nt fragments of the input sequences
- TETRA: calculates tetranucleotide frequencies of each input sequence
The algorithms take as input correctly-formatted FASTA multiple sequence files. All sequences for a single organism should be contained in only one sequence file. Although it is possible to provide new labels for each input genome for rendering graphical output, the names of these files are used for identification so it is best to name them sensibly.
Output is written to a named directory. The output files differ depending on the chosen ANI method.
- ANIm: MUMmer/NUCmer
.deltafiles, describing each pairwise sequence alignment. Output as tab-separated plain text format tables describing: alignment coverage; total alignment lengths; similarity errors; and percentage identity (ANIm). - ANIb and ANIblastall: FASTA sequences describing 1020nt fragments of each input sequence; BLAST nucleotide databases - one for each set of fragments; and BLASTN output files (tab-separated tabular format plain text) - one for each pairwise comparison of input sequences. Output as tab-separated plain text tables describing: alignment coverage; total alignment lengths; similarity errors; and percentage identity (ANIb or ANIblastall).
- TETRA: Tab-separated plain text files describing the Pearson correlations between Z-score distributions for each tetranucleotide in each input sequence (TETRA).
If graphical output is chosen, the output directory will also contain PDF, PNG and EPS files representing the various output measures as a heatmap with row and column dendrograms. Other output formats (e.g. SVG) can be specified with the --gformat argument.
Licensing
Unless otherwise indicated, all code is subject to the following agreement:
(c) The James Hutton Institute 2014-2019
(c) The University of Strathclyde 2019-present
Author: Leighton Pritchard
Contact: leighton.pritchard@strath.ac.uk
Address:
Leighton Pritchard,
Strathclyde Institute of Pharmacy and Biomedical Sciences
161 Cathedral Street
Glasgow
G4 0RE,
Scotland,
UK
The MIT License
Copyright (c) 2014-2019 The James Hutton Institute
Copyright (c) 2014-present The James Hutton Institute
Permission is hereby granted, free of charge, to any person obtaining a copy
of this software and associated documentation files (the "Software"), to deal
in the Software without restriction, including without limitation the rights
to use, copy, modify, merge, publish, distribute, sublicense, and/or sell
copies of the Software, and to permit persons to whom the Software is
furnished to do so, subject to the following conditions:
The above copyright notice and this permission notice shall be included in
all copies or substantial portions of the Software.
THE SOFTWARE IS PROVIDED "AS IS", WITHOUT WARRANTY OF ANY KIND, EXPRESS OR
IMPLIED, INCLUDING BUT NOT LIMITED TO THE WARRANTIES OF MERCHANTABILITY,
FITNESS FOR A PARTICULAR PURPOSE AND NONINFRINGEMENT. IN NO EVENT SHALL THE
AUTHORS OR COPYRIGHT HOLDERS BE LIABLE FOR ANY CLAIM, DAMAGES OR OTHER
LIABILITY, WHETHER IN AN ACTION OF CONTRACT, TORT OR OTHERWISE, ARISING FROM,
OUT OF OR IN CONNECTION WITH THE SOFTWARE OR THE USE OR OTHER DEALINGS IN
THE SOFTWARE.










